
PDF) A “three-eyed” infant: A case of partial facial duplication (diprosopus monocephalus triophthalmos)
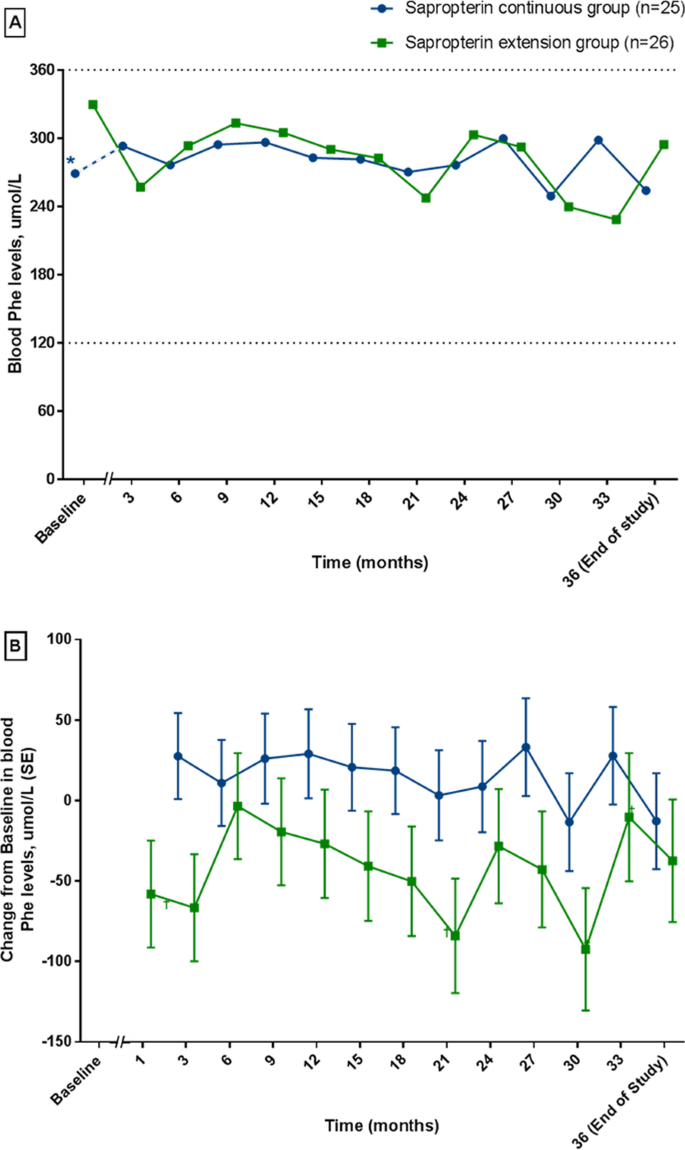
Long-term efficacy and safety of sapropterin in patients who initiated sapropterin at < 4 years of age with phenylketonuria: results of the 3-year extension of the SPARK open-label, multicentre, randomised phase IIIb
Cost-effectiveness of sapropterin dihydrochloride (Kuvan®) for the treatment of hyperphenylalaninaemia (HPA) in adults and paed
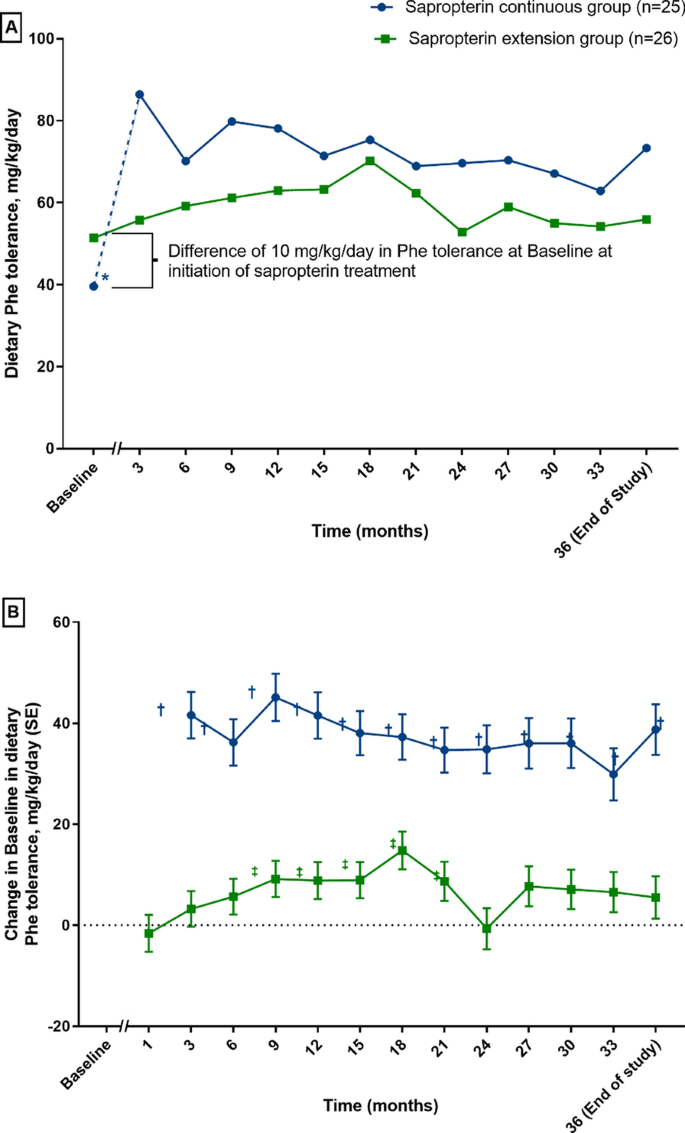
Long-term efficacy and safety of sapropterin in patients who initiated sapropterin at < 4 years of age with phenylketonuria: results of the 3-year extension of the SPARK open-label, multicentre, randomised phase IIIb












